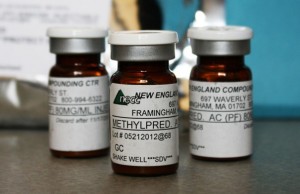
In October 2012, three batches of contaminated epidural steroid injections prepared by the
New England Compounding Center were linked to an
outbreak of
fungal meningitis. The outbreak led to
480 diagnoses and 33 deaths as of November 16, 2012, causing many to
call for increased regulations of compounding pharmacies.
The practice of
pharmaceutical compounding is relatively simple. It involves a pharmacist creating a medicine that is tailored to a patient’s needs. For example, pharmacists can create a prescription pill without a specific ingredient to serve a patient with an allergy, or they can convert that same pill into a liquid for easier consumption by some patients. The practice has become more common in recent years, and some see it as a
cost-saving alternative to large drug manufacturers.
Some critics are wary of this rise in popularity because most states
regulate compounding pharmacies at the state level, so the
U.S. Food and Drug Administration (FDA) has little or no authority over the regulations and procedures. The lack of consensus opinion on this issue is largely due to the Supreme Court’s decision in
Thompson v. Western States Medical Center, 535 U.S. 357 (2002).
In
Thompson, the Court ruled that certain limitations imposed on compacting under the
Food and Drug Modernization Act of 1997 (FDAMA), were unconstitutional. The Court did not rule, however, on whether those unconstitutional provisions were severable from the FDAMA, leaving open the question of whether the FDA’s remaining oversight of compounding pharmacies via the FDAMA was constitutional. Multiple
Circuit Courts of Appeals have addressed the issue, but there is still no consensus decision, and the Court has not addressed the question since. In short, compounding pharmacies exist in a legal grey area, with no uniform regulation.
In those states where the FDAMA is still in effect, the FDA must
consider a number of factors before deciding whether it will require compounding pharmacies to send their custom drugs in for FDA approval. In the states where the FDAMA is not in effect, such as
Massachusetts, the oversight of compounding pharmacies is left to state agencies, which is usually that state’s
Pharmacy Board.
Even in states where the FDAMA is not controlling, the FDA may still have some power. Under the
Food, Drug, and Cosmetic Act of 1938, the FDA can criminally prosecute those who cause an unapproved drug to enter interstate commerce. The FDA has never gone this far, as most pharmacies will stop their questionable production following an FDA letter threatening indictment.
The reason for this general lack of federal oversight is that compounding pharmacies have only recently begun to displace drug manufacturers. For the most part, compounding pharmacies are smaller
manufacturers, and the federal government typically defers to state regulation.
The result of the confusion over the FDAMA is that the FDA’s regulatory hands are tied, leaving state pharmacy boards to control the industry. This is not to say that this practice hasn’t been successful in the past. Apart from this latest outbreak, this practice has been relatively problem free. However, as the industry continues to grow, some type of uniform regulation should likely be enacted in order to make sure that an outbreak like this latest one does not happen again.
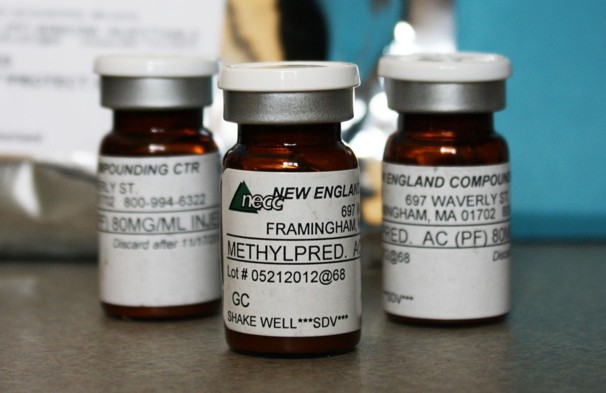
 In October 2012, three batches of contaminated epidural steroid injections prepared by the New England Compounding Center were linked to an outbreak of fungal meningitis. The outbreak led to 480 diagnoses and 33 deaths as of November 16, 2012, causing many to call for increased regulations of compounding pharmacies.
The practice of pharmaceutical compounding is relatively simple. It involves a pharmacist creating a medicine that is tailored to a patient’s needs. For example, pharmacists can create a prescription pill without a specific ingredient to serve a patient with an allergy, or they can convert that same pill into a liquid for easier consumption by some patients. The practice has become more common in recent years, and some see it as a cost-saving alternative to large drug manufacturers.
Some critics are wary of this rise in popularity because most states regulate compounding pharmacies at the state level, so the U.S. Food and Drug Administration (FDA) has little or no authority over the regulations and procedures. The lack of consensus opinion on this issue is largely due to the Supreme Court’s decision in Thompson v. Western States Medical Center, 535 U.S. 357 (2002).
In Thompson, the Court ruled that certain limitations imposed on compacting under the Food and Drug Modernization Act of 1997 (FDAMA), were unconstitutional. The Court did not rule, however, on whether those unconstitutional provisions were severable from the FDAMA, leaving open the question of whether the FDA’s remaining oversight of compounding pharmacies via the FDAMA was constitutional. Multiple Circuit Courts of Appeals have addressed the issue, but there is still no consensus decision, and the Court has not addressed the question since. In short, compounding pharmacies exist in a legal grey area, with no uniform regulation.
In those states where the FDAMA is still in effect, the FDA must consider a number of factors before deciding whether it will require compounding pharmacies to send their custom drugs in for FDA approval. In the states where the FDAMA is not in effect, such as Massachusetts, the oversight of compounding pharmacies is left to state agencies, which is usually that state’s Pharmacy Board.
Even in states where the FDAMA is not controlling, the FDA may still have some power. Under the Food, Drug, and Cosmetic Act of 1938, the FDA can criminally prosecute those who cause an unapproved drug to enter interstate commerce. The FDA has never gone this far, as most pharmacies will stop their questionable production following an FDA letter threatening indictment.
The reason for this general lack of federal oversight is that compounding pharmacies have only recently begun to displace drug manufacturers. For the most part, compounding pharmacies are smaller manufacturers, and the federal government typically defers to state regulation.
The result of the confusion over the FDAMA is that the FDA’s regulatory hands are tied, leaving state pharmacy boards to control the industry. This is not to say that this practice hasn’t been successful in the past. Apart from this latest outbreak, this practice has been relatively problem free. However, as the industry continues to grow, some type of uniform regulation should likely be enacted in order to make sure that an outbreak like this latest one does not happen again.
In October 2012, three batches of contaminated epidural steroid injections prepared by the New England Compounding Center were linked to an outbreak of fungal meningitis. The outbreak led to 480 diagnoses and 33 deaths as of November 16, 2012, causing many to call for increased regulations of compounding pharmacies.
The practice of pharmaceutical compounding is relatively simple. It involves a pharmacist creating a medicine that is tailored to a patient’s needs. For example, pharmacists can create a prescription pill without a specific ingredient to serve a patient with an allergy, or they can convert that same pill into a liquid for easier consumption by some patients. The practice has become more common in recent years, and some see it as a cost-saving alternative to large drug manufacturers.
Some critics are wary of this rise in popularity because most states regulate compounding pharmacies at the state level, so the U.S. Food and Drug Administration (FDA) has little or no authority over the regulations and procedures. The lack of consensus opinion on this issue is largely due to the Supreme Court’s decision in Thompson v. Western States Medical Center, 535 U.S. 357 (2002).
In Thompson, the Court ruled that certain limitations imposed on compacting under the Food and Drug Modernization Act of 1997 (FDAMA), were unconstitutional. The Court did not rule, however, on whether those unconstitutional provisions were severable from the FDAMA, leaving open the question of whether the FDA’s remaining oversight of compounding pharmacies via the FDAMA was constitutional. Multiple Circuit Courts of Appeals have addressed the issue, but there is still no consensus decision, and the Court has not addressed the question since. In short, compounding pharmacies exist in a legal grey area, with no uniform regulation.
In those states where the FDAMA is still in effect, the FDA must consider a number of factors before deciding whether it will require compounding pharmacies to send their custom drugs in for FDA approval. In the states where the FDAMA is not in effect, such as Massachusetts, the oversight of compounding pharmacies is left to state agencies, which is usually that state’s Pharmacy Board.
Even in states where the FDAMA is not controlling, the FDA may still have some power. Under the Food, Drug, and Cosmetic Act of 1938, the FDA can criminally prosecute those who cause an unapproved drug to enter interstate commerce. The FDA has never gone this far, as most pharmacies will stop their questionable production following an FDA letter threatening indictment.
The reason for this general lack of federal oversight is that compounding pharmacies have only recently begun to displace drug manufacturers. For the most part, compounding pharmacies are smaller manufacturers, and the federal government typically defers to state regulation.
The result of the confusion over the FDAMA is that the FDA’s regulatory hands are tied, leaving state pharmacy boards to control the industry. This is not to say that this practice hasn’t been successful in the past. Apart from this latest outbreak, this practice has been relatively problem free. However, as the industry continues to grow, some type of uniform regulation should likely be enacted in order to make sure that an outbreak like this latest one does not happen again.

